Table of Contents
Have you ever looked at a chemistry formula and thought, “What does that even mean?” If you’ve seen hcooch ch2 h2o and felt confused, you’re not alone. Chemistry can seem tricky at first, but once you break it down using easy words and examples, it starts to make more sense. This article is here to do exactly that. We’re going to explore this simple-looking formula step-by-step, so you can understand what’s happening and why it matters.
The focus keyword hcooch ch2 h2o may seem like a random mix of letters and numbers, but it actually points to a key reaction from organic chemistry. We’ll explain what each part means, the science behind it, and why it’s important in everyday life. You’ll learn about esters, hydrolysis, and chemical equations—all in a way that even a fifth-grader can follow. Along the way, we’ll include real-life uses, simple diagrams, and fun facts to keep things interesting.
Ready to turn this from confusing to clear? Let’s dive into the world of HCOOCH2 H2O and see just how easy chemistry can really be.
What Does HCOOCH₂ Represent?
Let’s start with understanding what HCOOCH₂ really is. This chemical is called methyl formate. Yes, it might sound like a name out of a science movie, but in everyday terms, it’s an ester. Esters are a kind of chemical that often smells sweet or fruity. They frequently show up in perfumes, flavorings, and even in bananas and apples!
In this formula, “HCOO” stands for formic acid (a simple acid found in ants!), and “CH₂” means that part of the molecule is bonded with a single carbon and two hydrogen atoms. Put together, HCOOCH₂ forms methyl formate, which is made from formic acid and methanol reacting together.
In short, it’s a small, simple ester with a very interesting role in chemistry. Learning about what this little molecule can do helps you better understand bigger concepts in science.
What Happens When You Add H₂O?
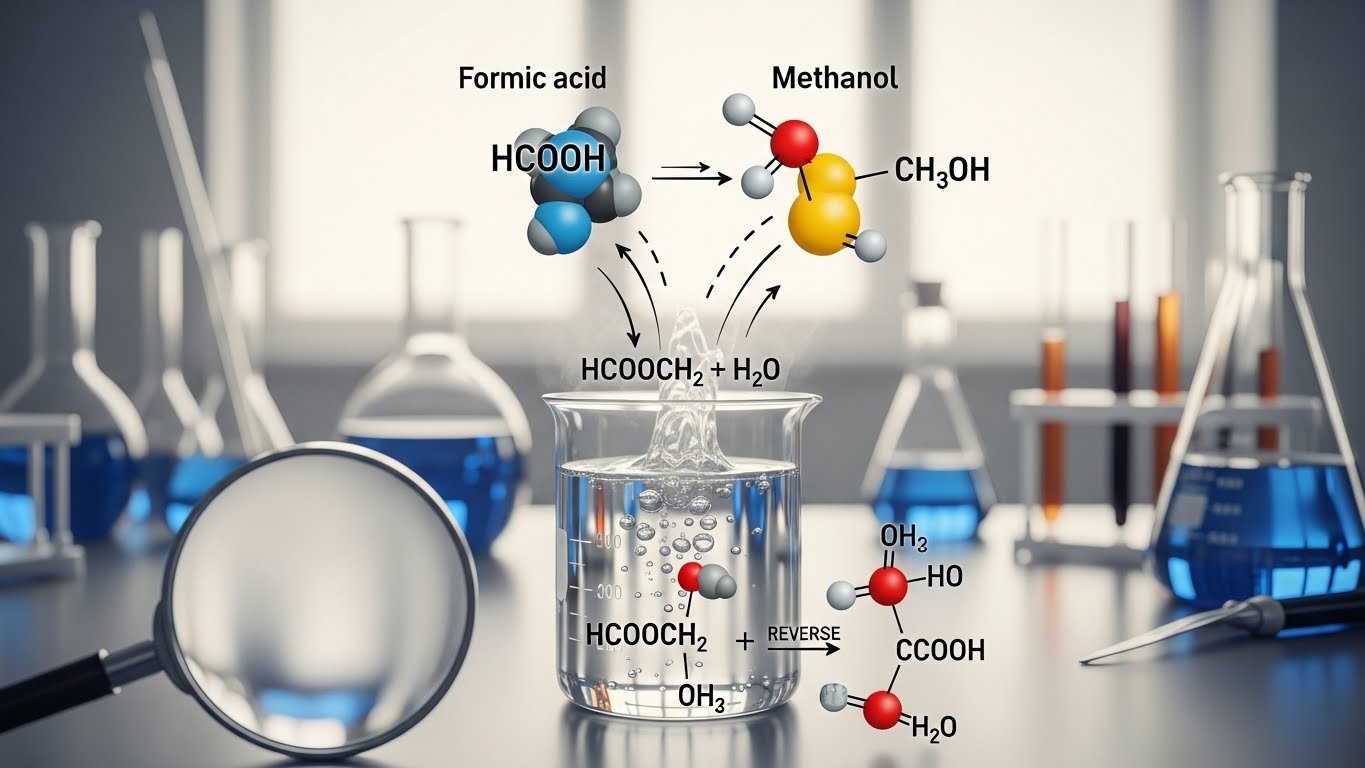
So what happens when you mix HCOOCH₂ with H₂O (which is just water)? This is where the science gets exciting. The process is called hydrolysis. That’s just a fancy word that means “to break something apart using water.”
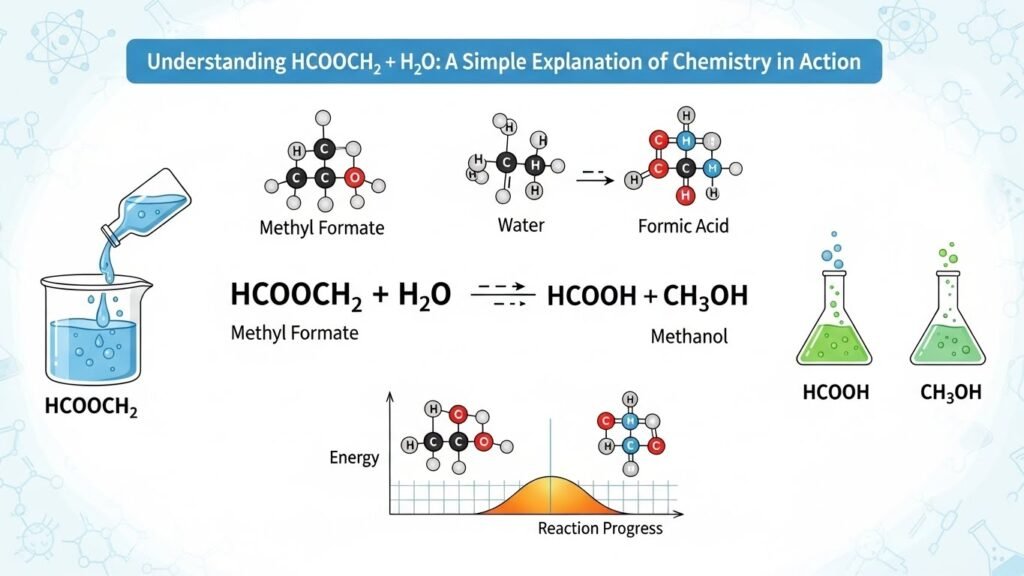
When water is added to an ester like methyl formate, the molecule splits apart. In simple terms, adding water “breaks” the ester into two pieces—one is an acid (formic acid), and the other is an alcohol (methanol). It’s a chemical reaction that reverses the way the ester was created in the first place.
This reaction is important not only because of what it does but also because of where it happens—our bodies, factories, labs, and even the environment. It helps show how water can change and break down molecules.
Let’s Break It Down: The Chemical Reaction
The equation for this reaction is:
hcooch ch2 h2o → HCOOH + CH₃OH
If that looks like a lot, don’t worry—we’ll explain. Here’s what each part stands for:
- HCOOCH₂ is methyl formate, a type of ester.
- H₂O is water.
- HCOOH is formic acid.
- CH₃OH is methanol, also known as wood alcohol.
So, the ester reacts with water and breaks down into one acid (formic acid) and one alcohol (methanol). This is a classic example of ester hydrolysis.
Understanding reactions like this helps students, teachers, and scientists predict how substances behave in different environments. It can even help in food science and medicine.
What Is Ester Hydrolysis?
We’ve used the word “hydrolysis” a few times, so let’s focus on it now. In chemistry, hydrolysis means “splitting with water.” When it comes to ester hydrolysis, it means using water to break an ester into its original parts—an acid and an alcohol.
In this case, we’re taking the ester methyl formate (HCOOCH₂) and breaking it using water (H₂O). The result is formic acid and methanol. This is a reversible reaction, meaning that under the right conditions, you could reverse it and put the pieces back together to form the ester again.
Hydrolysis is important in nature and industry. Our bodies use hydrolysis to digest fats and proteins. In factories, hydrolysis can help break down plastics or make flavors and fragrances.
Fun Fact: Esters Smell Like Fruit!
Did you know that many esters smell like fruits or flowers? Methyl formate (HCOOCH₂) has a slightly fruity scent. That’s why esters are often found in perfumes, air fresheners, and food flavorings.
In fact, many natural fruits contain esters. That banana smell? That’s due to isoamyl acetate, which is an ester. Pineapple? That’s ethyl butyrate, another ester. Methyl formate might not be as well-known in foods, but it’s still a sweet-smelling part of the ester family.
So yes, the same group of chemicals you’re learning about in this reaction—the HCOOCH2 H2O hydrolysis—can also be responsible for the delicious smells you find in your kitchen or garden.
Where Is This Reaction Used in Real Life?
You might be wondering, why should I care about methyl formate reacting with water? Well, this reaction isn’t just for science class. It has real-world uses!
In chemical factories, engineers use reactions like this to carefully control how substances are made or broken down. In pharmaceuticals, understanding hydrolysis helps doctors know how some drugs break down in your body. In environmental science, this reaction helps explain how water in rivers or rain breaks down pollutants.
Even the perfume and flavor industries use knowledge of HCOOCH2 H2O to create better-smelling products. So, even though the equation looks like a mystery at first, it actually connects to many parts of everyday life.
Why Water Makes It All Possible
Water is more powerful than it looks. In this reaction, H₂O isn’t just a filler—it’s the active player. It breaks the ester molecule apart by attacking the bond between the acid and alcohol parts.
Here’s how it works in simple terms: water “splits” into H⁺ (a hydrogen ion) and OH⁻ (a hydroxide ion). These two parts sneak into the ester and help it fall apart. One grabs onto the acid side, and the other grabs onto the alcohol side.
This is why water is often called the “universal solvent.” It does more than just dissolve things—it participates in important reactions like HCOOCH2 H2O hydrolysis, making chemistry happen.
What Is Methanol and Why Does It Matter?
Let’s take a closer look at one of the products of this reaction: methanol (CH₃OH). It’s also called “wood alcohol” and was once made by heating wood. Today, it’s used as fuel, in antifreeze, and to create other chemicals.
But methanol can be dangerous. Drinking even small amounts can cause serious poisoning. That’s why it’s important to understand how it’s made and handled. In this case, the hydrolysis of methyl formate produces methanol. That’s why safety is super important when doing chemical reactions like this.
In controlled environments, scientists use this reaction to safely make or study methanol. Understanding this helps make workplaces and products safer for everyone.
A Closer Look at Formic Acid
The other product of this reaction, formic acid (HCOOH), is also interesting. It’s the simplest type of carboxylic acid and can be found in the stings of ants and bees! That burning feeling? It’s because of formic acid.
In industry, formic acid is used in leather tanning, rubber production, and as an antibacterial agent in livestock feed. It’s powerful but useful. Knowing that this acid can come from breaking apart something that smells nice—like methyl formate—shows how chemistry can surprise us.
This is another reason people search “HCOOCH2 H2O.” They want to see how something small and sweet can split apart into something sharp and strong.
Is This a Reversible Reaction?
Yes, the hydrolysis of HCOOCH₂ is a reversible reaction. That means the products—formic acid and methanol—can go back and form methyl formate again under the right conditions.
In chemistry, reversible reactions can go forward and backward. In labs, heating or adding a catalyst can push a reversible reaction one way over the other. This means we can control which way the arrow points in the equation based on temperature, pressure, or pH level.
Understanding this helps scientists recycle materials, design drug processes, and study how living things work. It’s like a chemical see-saw—and discovering how to tilt it just right is what makes chemistry so cool.
How This Knowledge Helps Students
If you’re a student, learning reactions like HCOOCH2 H2O → HCOOH + CH3OH helps build your foundation in chemistry. It teaches you about molecular structures, how bonds form and break, and how water plays a role in more than just hydration.
Knowing these basics sets you up for higher-level studies, including organic chemistry, biology, and even medicine. Plus, it boosts your critical thinking skills. You learn to look beyond the surface of a formula and see the science working underneath.
So, even if you’re just starting your science journey, this simple reaction can be your first step into the amazing world of chemistry.
FAQs
1. What kind of reaction is hcooch ch2 h2o?
It’s a hydrolysis reaction, where water helps break an ester into an acid and alcohol.
2. What does HCOOCH2 stand for?
HCOOCH2 is methyl formate, an ester made from formic acid and methanol.
3. What are the products of this reaction?
The reaction creates formic acid (HCOOH) and methanol (CH₃OH).
4. Is this reaction reversible?
Yes, it can go both forward and backward under the right conditions. It’s a reversible reaction.
5. Where is this reaction used in real life?
It’s used in chemistry labs, factories, medicine, flavor-making, and environmental science.
6. Is methyl formate safe?
In small amounts and controlled settings, yes. But it can be flammable, and methanol (from it) is toxic, so precautions are needed.
Conclusion
Who knew HCOOCH2 H2O could teach us so much? This tiny formula holds powerful lessons. It shows us how esters break apart, how water helps reactions, and how acids and alcohols are formed. It’s more than just letters and numbers—it’s a real science story that happens in labs, nature, and even your own body. Whether you’re a student, a curious reader, or a science fan, understanding these reactions helps you see the world in a smarter way. Science doesn’t need to be scary. When we break it into small parts—just like water breaks the ester—everything becomes clearer.